Abbott's FreeStyle® Libre 2 iCGM Cleared in U.S. for Adults and Children with Diabetes, Achieving Highest Level of Accuracy and Performance Standards

FDA clears Abbott's FreeStyle Libre 2 system in U.S. for adults and children (ages 4 and older) with diabetes, the only iCGM with unsurpassed 14-day accuracy that measures glucose every minute with optional real-time alarms.
/PRNewswire/ -- Abbott (NYSE: ABT), the worldwide leader in continuous glucose monitoring (CGM), announced today the U.S. Food and Drug Administration (FDA)

Abbott's FreeStyle® Libre 2 iCGM Cleared in U.S. for Adults and Children with Diabetes, Achieving Highest Level of Accuracy and Performance Standards

Monitoring Technologies- Continuous Glucose Monitoring, Mobile Technology, Biomarkers of Glycemic Control - Endotext - NCBI Bookshelf
Abbott Releases the FreeStyle Libre 2 iCGM - Children with Diabetes

Abbott's FreeStyle® Libre 2 iCGM Cleared in U.S. for Adults and Children with Diabetes, Achieving Highest Level of Accuracy and Performance Standards

Advances in Continuous Glucose Monitoring and Integrated Devices for Management of Diabetes with Insulin-Based Therapy: Improvement in Glycemic Control
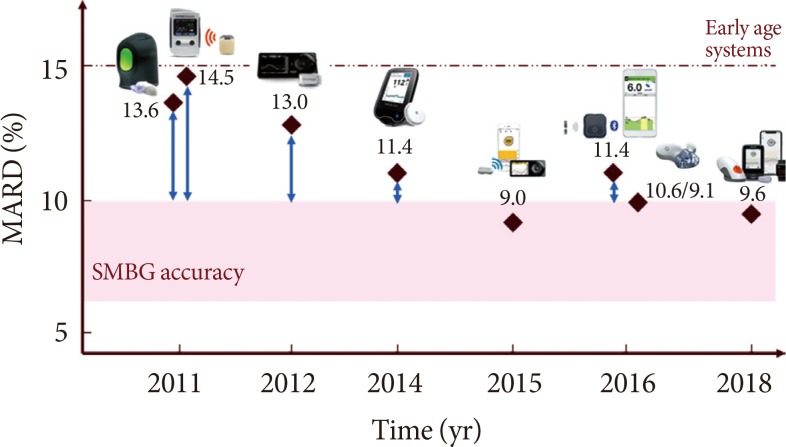
Continuous Glucose Monitoring Sensors for Diabetes Management: A Review of Technologies and Applications

Abbott, Medtronic go head-to-head at ADA conference - Page 2 of 9 - Medical Design and Outsourcing

FreeStyle Libre 2 Continuous Glucose Monitor
2 Million Patients And Beyond: Abbott's FreeStyle Libre 2 Cleared In The U.S. For Adults And Children With Diabetes

Improving Equitable Access to Continuous Glucose Monitors for Alabama's Children with Type 1 Diabetes: A Quality Improvement Project

Top 10 Companies in Over The Counter (OTC) Tests Market in 2023
Abbott's FreeStyle® Libre 2 iCGM Cleared in U.S. for Adults and Children with Diabetes, Achieving Highest Level of Accuracy and Performance Standards - Jun 15, 2020

FreeStyle Libre 2 Sensor
Freestyle Libre - Continuous Glucose Monitoring - Diabetes Monitors







